Silicon nanowires are studied in many projects related to the future of electronics, photovoltaic conversion, thermoelectricity, sensors etc. But their electronic properties are still not well-known. In particular, the influence of the catalyst needed to fabricate them has never been rigorously evaluated. At SiNaPS, we have shown that the radiative recombination properties of nanowires depends predominantly on surface passivation and not on the catalyst.
Silicon nanowires (Figure 1) are fabricated by vapor-liquid-solid catalytic growth. The catalyst is a gold nanoparticle, and the nanowire grows from a melted silicon gold alloy. One possible issue is that a gold atom in a silicon matrix is a defect that can alter the electronic properties. How can this be studied?

Figure 1: Nanowire forest and a closer view upon which the passivating silicon oxide layer can be seen.
Photoluminescence –
i.e., light emission induced by a laser excitation – permits characterizing the properties of charge carriers in a semiconductor (see inset) without the constraints associated with contacts in electrical measurements. We have measured the surface recombination velocity by time-resolved photoluminescence. For this purpose, we use a pump-probe technique. The excitation beam (the pump) creates excitons. The probe measures the photoluminescence after a chosen delay. By varying this delay, we have access to both the photoluminescence intensity decay time Τ and to the surface recombination velocity (SRV) of the carriers, which is a measure of material quality.
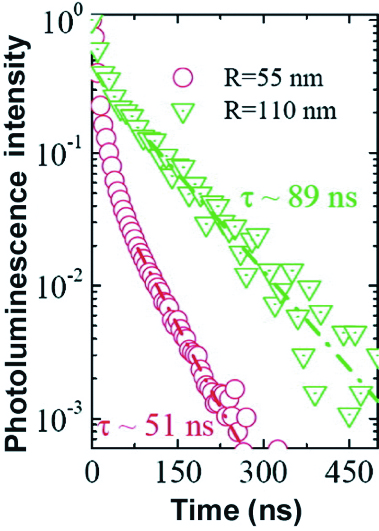
We first applied our technique to high quality Si thin films. Photoluminescence decay is faster for thinner layers. This suggests that recombination processes take place at the interface between silicon and silicon oxide. The SRV also depends on the passivation treatment that we use. We then studied our nanowires. For one kind of passivation, ô increases when the diameter increases (Figure 2). For a given nanowire diameter, the SRV increases when the passivated oxide layer is thicker. By combining all of this data (Figure 3), we find that the volume parameters of the nanowires are identical to those of the best silicon crystals! This shows that, once passivated, the nanowires are not degraded by the catalyst used to fabricate them: what matters is the quality of the surface passivation.
|
Figure 2 | |
Figure 3 |
| | |
 | |
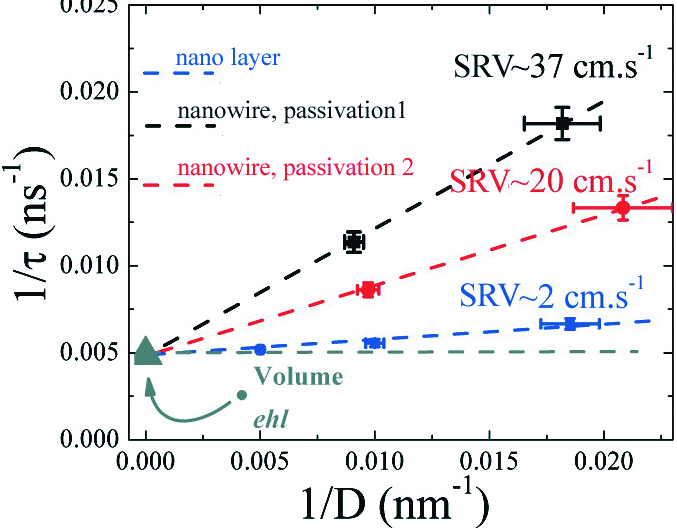 |
Figure 2: Photoluminescence intensity decay for two assemblies of nanowires with different diameters, but passivated in the same way. Measurements were taken at 10 K.
Figure 3: Inverse of the decay time as a function of the inverse of the diameter. The slope gives the surface recombination velocity (SRV) of the electron-hole pairs directly, while the ordinate at origin gives the volume recombination time.
Electron-hole drops
When a laser with a wavelength larger than the bandgap is shone on a semiconductor, bound electron-hole (e-h) pairs are created, and called excitons. When the density of such excitons is too large, the e-h bonds break and the exciton system becomes a plasma of dissociated electrons and holes with strong Coulomb interactions. At low temperature ( below 25 K for Si ), this condensed phase becomes a liquid. In a thin silicon layer, this exciton liquid (EHL) takes the shape of a drop in equilibrium with its vapor. The radius of the drop is of the order of several hundred nanometers. In contrast, for a passivated nanowire (less than 100 nm in diameter for a length of several micrometers), this EHL phase is present in practically all the available space. The particular electronic structure of silicon allows for photoluminescent observation of this original phase, thanks to the relatively long lifetimes (~1μs) of excitons, as compared to other semiconductors.